For the first time in 30 years, the American Pediatrics Academy (doctors who care for children’s health) has substantially separated from the government’s recommendations in vaccines.
The new recommendations of pediatricians about the Coronavirus, published on Tuesday, arrive in a tumultuous year for public health: skeptics about vaccines have come to power in the new Government of Donald Trump, and the guidelines of the administration have become increasingly confusing.
This is not going to help, James Campbell, vice president of the Infectious Diseases Committee of the Academy of Pediatricians, admitted “will be confusing, but our opinion is that we must make the right decision to protect children.”
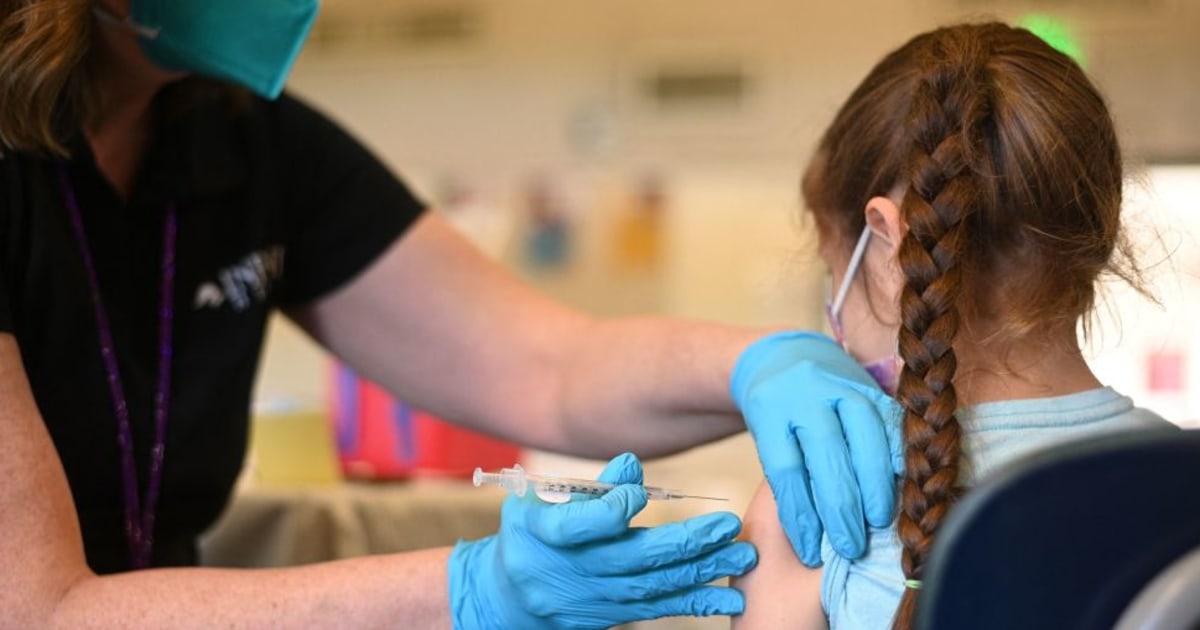
Pediatricians strongly recommend vaccines against the coronavirus for children from 6 months to 2 years, and also for the elderly if fathers and mothers want them to be immunized.
This differs from the guidelines established by the Secretary of Health, Robert F. Kennedy Jr., who does not recommend vaccines for healthy children of any age, but ensures that children can be vaccinated after consulting their doctors.
Children between 6 months and 2 are at a high risk of suffering a serious Covid-19 disease, and it is important that the recommendations continue to emphasize the need for them to be vaccinated, said Campbell, an expert in infectious diseases at the University of Maryland.
Vaccines are also recommended for older children suffering from chronic pulmonary diseases or other conditions that expose them to a greater risk of serious diseases, according to pediatricians.
The Pediatric Academy, founded 95 years ago, has issued recommendations on children’s vaccination since the 1930s. In 1995, it synchronized its advice with the recommendations of the Centers for Disease Control and Prevention (CDC), under the Department of Health.
Since then, there have been some small differences between the recommendations of the pediatricians and those of the CDC. For example, it advises that children are vaccinated against human papillomavirus (HPV) from 9 years; CDC say that is fine, but they have emphasized vaccination at 11 and 12.
In 30 years, this is the first time that the recommendations differ “significantly or substantially,” Campbell said.
Until recently, the CDC, following the recommendations of infectious diseases, recommended that all 6 months receive an annual reinforcement dose against COVID-19.
(Are you afraid of needles? This nasal spray vaccine is ready to use at home)
However, in May, the Health Department announced that vaccines against COVID-19 for healthy children and pregnant women were no longer recommended. Days later, the CDC said a statement in which they indicated that healthy children could be vaccinated, but that it was no longer a “mandatory” recommendation.
The idea that healthy older children can omit the reinforcement doses against COVID-19 have been being developed among public health experts. As the pandemic has been referring, several specialists have increasingly discussed the possibility of focusing vaccination efforts on people 65 years of age or older, who are among those who run the greatest risk of death and hospitalization.
In June, a panel of CDC experts met to broadcast recommendations on autumn vaccines. Among the options that was considering was recommending immunizations for high -risk groups, but leaving people with less danger to vaccinating.
But the Department of Health overlooked the group, and decided to dismiss the panel of 17 members and name a smaller one that included skeptics about vaccines. Later, it excluded the Pediatric Academy, the American Medical Association and other first -class medical organizations to collaborate with the advisors to establish vaccination recommendations.
The new vaccine panel has not voted recommendations on the Vaccine against COVID-19 yet. He supported the recommendation to continue with flu vaccines in autumn, but also made a decision that meant another remarkable difference with pediatricians: he said that people should only receive flu vaccines that are presented in unique dose containers and that they do not contain the thymatory preservative.
Pediatricians claim that there is no evidence that the preservative is detrimental and recommended that doctors use any authorized flu vaccine product that is suitable for the patient.