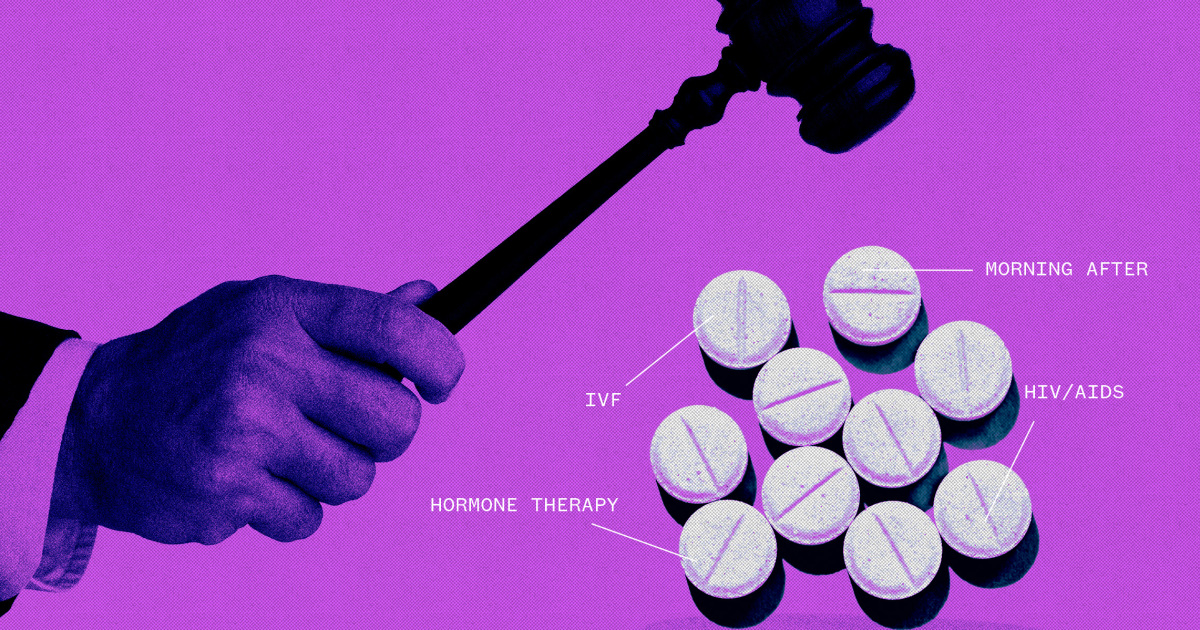
WASHINGTON — Vaccines, birth control pills, hormone therapies and fertility drugs would be the subject of new litigation if the Supreme Court upholds a challenge to the abortion pill mifepristone, pharmaceutical industry experts warn.
Next Tuesday, when the court considers whether to revoke the decisions of the Food and Drug Administration (FDA) that facilitated access to mifepristone, not only will access to this drug that is used to the majority of abortions in the entire country.
The pharmaceutical industry has expressed alarm and has told judges and anyone who will listen that giving federal judges the power to overturn the agency's scientific conclusions on health and safety would create chaos in the sector.
Litigation is likely over other drugs, both those approved and those about to be approved, about which people have strong opinions.
If anti-abortion groups win, “anyone with an ideological disagreement, coupled with a judge without scientific training, could challenge the authority of the FDA,” said Amanda Banks, a doctor and businesswoman who signed a document along with dozens of other executives and pharmaceutical companies. who support the FDA.
The main pharmaceutical industry group, PhRMA, also submitted a brief in support of the Government.
Some activists have long attacked certain vaccines, alleging without evidence that they can cause autism. During the COVID-19 pandemic, vaccine skeptics raised largely unfounded concerns that the vaccines were unsafe.
Anti-abortion activists, some of whom oppose all contraceptives, have long opposed the morning-after pill, viewing it as similar to abortion despite evidence suggesting otherwise.
LGBTQ activists have called on the FDA to specifically approve the use of hormone therapies for gender-affirming treatments. Groups opposed to transgender youth receiving gender-affirming treatments have asked the FDA to prevent puberty blockers from being prescribed to minors.
Other drugs that could be questioned include those developed from embryonic stem cells, drugs to treat HIV/AIDS and fertility drugs used in in vitro fertilization, industry experts and other FDA supporters say. .
Business owners are also concerned that a ruling against the government could limit innovation and deter investors from a sector that invests billions of dollars in initial research and development to bring drugs to market.
“What worries me most is the precedent it sets… it could have a chilling effect on investors coming into our business and investing in our innovative companies,” said Paul Hastings, an industry veteran who is Nkarta's CEO. Therapeutics and that he signed the same document as Banks.
Hastings and others stressed that the FDA is considered the “gold standard” of pharmaceutical regulation around the world. For investors, FDA approval is the final part of a rigorous and expensive process. According to Hastings, only 1 in 10 drugs in development end up being marketed.
Investors might look for safer bets if FDA approval only leads to continued litigation, he added.
In court papers, the FDA itself said that no court has ever restricted access to an approved drug “by questioning the FDA's expert judgment about the conditions necessary to ensure the safe use of that drug.”
When the agency began the process of lifting restrictions on the drug in 2016, its actions were “supported by a comprehensive review of a record that included dozens of scientific studies and decades of safe use of mifepristone by millions of women,” the report said. written.
The plaintiffs, doctors and other medical professionals who oppose abortion, contend that the FDA failed to take safety concerns into account when it lifted restrictions on mifepristone.
Erin Hawley, an attorney for the conservative Christian group Alliance Defending Freedom, which is defending the case on behalf of the plaintiffs, rejected the idea that the case could have such far-reaching effects, calling it a “red herring.”
He said in an interview that the FDA has not identified any other drug approvals that were threatened.
“The reason is that what the FDA did in deciding to eliminate long-standing protections for women who decide to take mifepristone is quite extraordinary,” she added.
The group does not plan to challenge any other drug approvals, a spokeswoman said.
The Supreme Court has a 6-3 conservative majority that has been hostile to abortion rights, as clearly illustrated by the 2022 ruling that overturned the landmark Roe v. Wade.
The legal issue in Tuesday's case centers not on abortion itself, but on whether the FDA followed correct processes in relaxing restrictions on mifepristone.
The plaintiffs filed the litigation in federal court in Texas, where the case was guaranteed to be assigned to Matthew Kacsmaryk, a conservative judge appointed by President Donald Trump.
Kacsmaryk issued a sweeping ruling in April last year invalidating the FDA's approval of the drug decades ago.
That ruling was put on hold by the Supreme Court and has subsequently been reduced by the United States Court of Appeals for the Fifth Circuit.
The original approval of mifepristone in 2000 is not in question before the judges. The case focuses on FDA actions starting in 2016 that made the pill easier to access, including the initial 2021 decision that made it available by mail, which was finalized last year.
The judges will also examine 2016 decisions to extend the period in which mifepristone can be used to terminate pregnancies from seven to 10 weeks of gestation and reduce the number of in-person visits for patients from three to one. In another action in 2016, the FDA changed the dosing regimen, finding that a lower dose of mifepristone was sufficient.
The FDA-approved regimen for medical abortion includes two drugs: mifepristone, which blocks the hormone progesterone, and misoprostol, which induces contractions.
The fact that the judges left Kascmaryk's sentence on hold could be a sign that the majority is inclined to reject the challenge. Only two of the nine justices, conservatives Clarence Thomas and Samuel Alito, opposed that decision.
It is possible that the court will resolve the case without delving into the complicated legal questions surrounding the FDA approval process. The Government has vigorously argued that the doctors and others who brought the lawsuit lack standing because they cannot demonstrate any harm that could be attributed to the FDA's decisions.
Doctors themselves do not prescribe mifepristone, but suggest they are harmed because they may be forced to treat patients who have taken the pill and have serious side effects. Because they oppose abortion, any action they are forced to take to help a woman complete the process would make them complicit, the plaintiffs argued in court papers.
FDA attorneys wrote in the government's brief that plaintiffs can, at best, point to a “hypothetical scenario,” which is not enough to establish standing.
The challengers, the document stated, “cannot identify a single case in which any of their members have been forced to provide such treatment.”